Big Give 2025 - From Lab to People: Removing Barriers to Treatment
In 2024, over 300 Friends of Ataxia UK came together to raise a wonderfully festive total of over £57,500 during the Christmas Challenge week, helping fund our work with pharmaceutical companies and government agencies to bring drugs to the UK.
Your support in 2024 enabled us to pass a number of significant milestones this year as we continue to navigate the drug approval process.
The Big Give 2025: 2–9 December
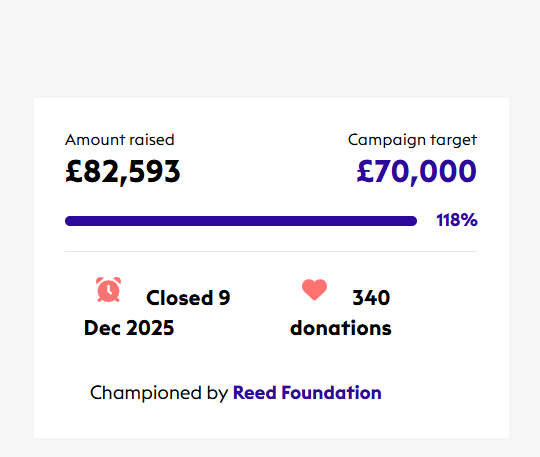
From 2–9 December, the ataxia community came together for The Big Give 2025, and the result was historic. Thanks to your incredible generosity, we raised over £82,000, the highest total ever for our campaign.
November 2025: At PMQs in the House of Commons, Jonathan Brash MP raised the urgent need for early access to treatment for people affected by ataxia. His statement prompted a response from the Deputy Prime Minister, who agreed to a meeting with Ataxia UK. This marks an important step forward in highlighting the issue at the national level.
September 2025: We submitted a second formal letter to the Department of Health and Social Care, pressing for a compassionate access programme & seeking an urgent meeting. We encouraged ataxians everywhere to forward the letter to their constituency MP, so that we could build a coalition of parliamentarians and policymakers willing to support our campaign. We also issued a press release to national media to highlight the injustice and keep the issue in the public eye.
June 2025: We published and sent an open letter to the Health Secretary, which was signed by over 10,000 supporters. Ataxia UK CEO Sue Millman and Professor Paola Giunti presented the case at the All-Party Parliamentary Group on Rare Diseases.
January 2025: We submitted a detailed report to NICE as part of the evaluation process for Omav.
This year's Big Give campaign is a continuation and expansion of this work. When progress towards treatments stalls, we come together. Not just for the drugs we cannot access now, but to improve the approval processes for the drugs to come. We want to use our position to advocate for the ataxia community, expand our engagement with policy stakeholders & encourage change.
The process of drug approval for use on the NHS hasn’t made Omav, the one licensed treatment for FA patients, available. Without change, the process could delay future drugs. People with rare conditions in the UK must not be kept waiting unnecessarily for treatments that are available in other countries. We must seize this key moment.